Physical Chemistry Chemical Physics – Chiara Negri, Andrea Martini, Gabriele Deplano, Kirill A. Lomachenko, Ton V. W. Janssens, Elisa Borfecchia, Gloria Berlier and Silvia Bordiga.
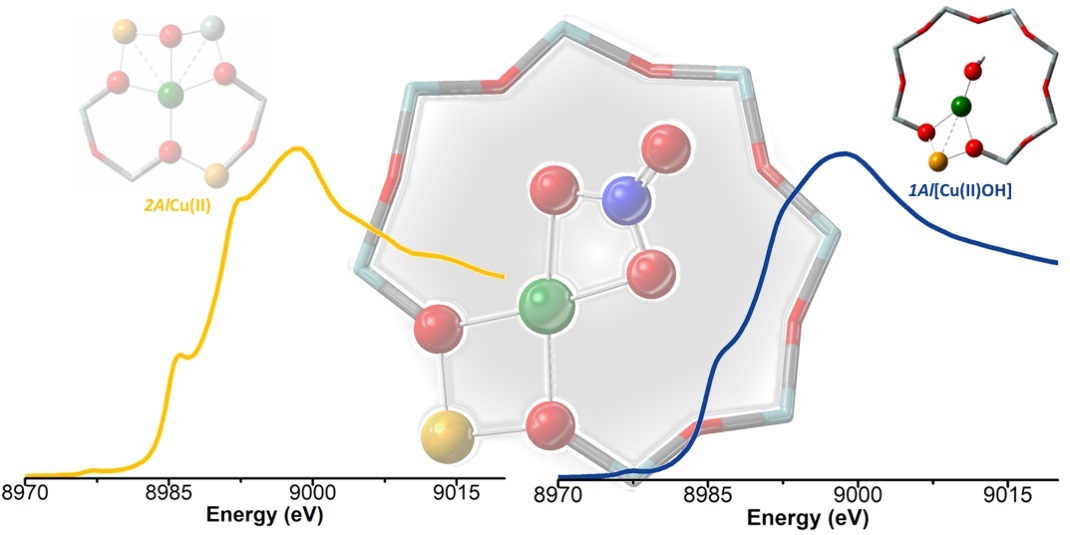
The speciation of framework-interacting CuII sites in Cu-chabazite zeolite catalysts active in the selective catalytic reduction of NOx with NH3 is studied, to investigate the influence of the Al content on the copper structure and their reactivity towards a NO/O2 mixture. To this aim, three samples with similar Cu densities and different Si/Al ratios (5, 15 and 29) were studied using in situ X-ray absorption spectroscopy (XAS), FTIR and diffuse reflectance UV-Vis during pretreatment in O2 followed by the reaction. XAS and UV-Vis data clearly show the main presence of Z2CuII sites (with Z representing a framework negative charge) at a low Si/Al ratio, as predicted. EXAFS wavelet transform analysis showed a non-negligible fraction of proximal Z2CuII monomers, possibly stabilized into two 6-membered rings within the same cage. These sites are not able to form Cu-nitrates by interaction with NO/O2. By contrast, framework-anchored Z[CuII(NO3)] complexes with a chelating bidentate structure are formed in samples with a higher Si/Al ratio, by reaction of NO/O2 with Z[CuII(OH)] sites or structurally similar mono- or multi-copper Zx[CuIIxOy] sites. Linear combination fit (LCF) analysis of the XAS data showed good agreement between the fraction of Z[CuII(OH)]/Zx[CuIIxOy] sites formed during activation in O2 and that of Z[CuII(NO3)] complexes formed by reaction with NO/O2, further confirming the chemical inertia of Z2CuII towards these reactants in the absence of solvating NH3 molecules.
This article is part of the themed collection: 2021 PCCP HOT Articles!
Click here for the complete article!